Cardiovascular disease remains the world’s leading cause of death, claiming close to 20 million lives each year, with cases continuing to rise across both high- and low-income countries. Despite decades of therapeutic advances, many patients still experience suboptimal outcomes, adverse drug reactions, or ineffective treatment due to variability in how individuals respond to cardiovascular medications.
Pharmacogenomics offers a compelling solution by using genetic insights to guide drug selection and dosing, moving cardiovascular care beyond one-size-fits-all prescribing. As highlighted in recent advances in cardiovascular pharmacogenomics, the scientific evidence supporting gene-guided therapy is growing, yet real-world clinical adoption remains uneven.
This is where artificial intelligence may play a critical enabling role. By helping clinicians interpret complex genetic data and integrate it into routine workflows, AI could accelerate the implementation of precision pharmacogenomics at scale. However, realizing this potential will depend on foundational elements such as data quality, clinical training, and robust digital infrastructure.
This blog explores the key principles, clinical applications, challenges, and future directions of cardiovascular pharmacogenomics, drawing on the latest insights from Dr. Omar Elsaka’s Progress in Cardiovascular Pharmacogenomics and complementary research in the field.
What Is Pharmacogenomics?
At its core, pharmacogenomics (PGx) studies how genetic variation affects an individual’s response to medications. Human DNA differs from person to person, and many of these variations — especially single nucleotide polymorphisms (SNPs) — can influence how a drug is absorbed, metabolized, transported, or even how its target responds physiologically.
Read our blog on expanding access to pharmacogenetic testing.
The outcome? Two patients on the same drug might experience very different results: one may benefit greatly, another may see no effect, and a third might even suffer harmful side effects.
Why it matters:
- ≥ 98% of people carry at least one pharmacogenomic variant.
- 60% of patients in primary care receive prescriptions affected by pharmacogenomic factors.
These figures highlight how widespread genetic influences on drug response truly are — especially in cardiovascular medicine, where common drugs like clopidogrel, warfarin, and statins are impacted.
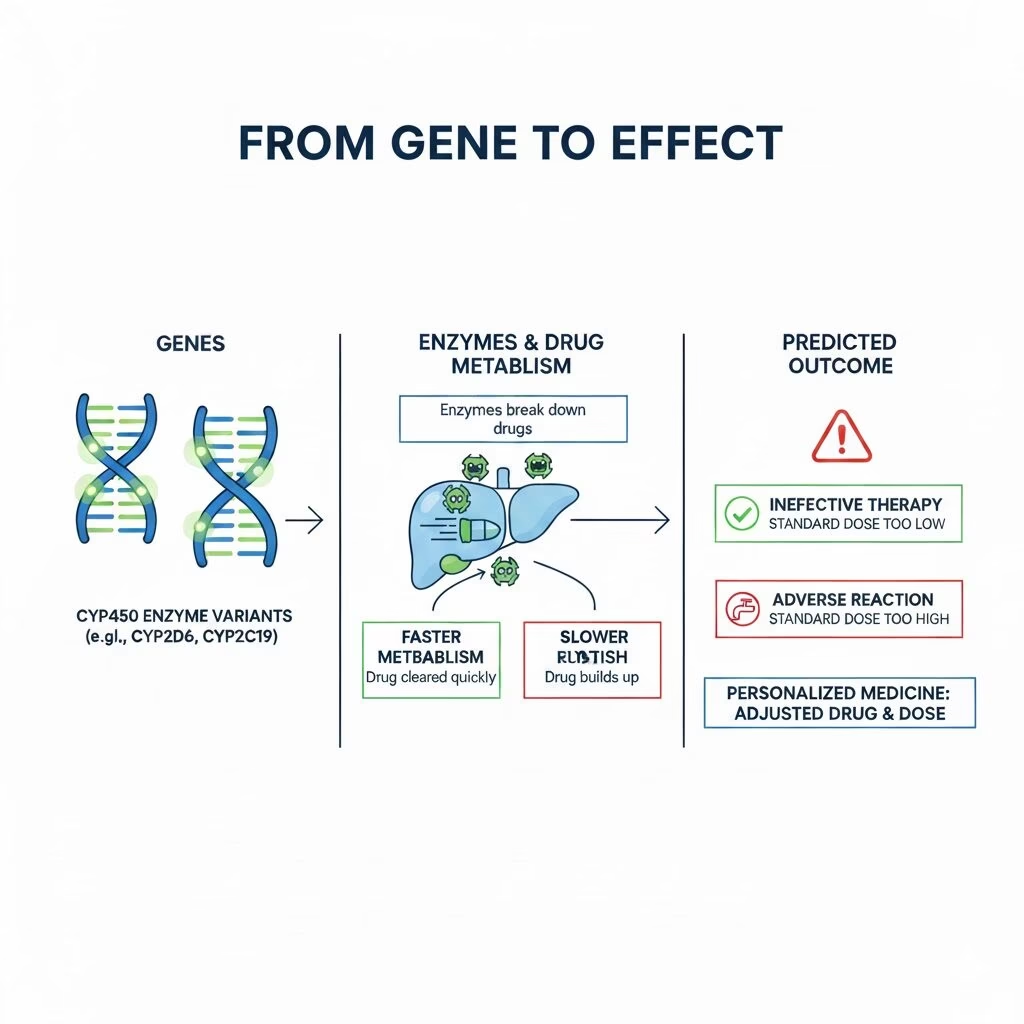
How Pharmacogenomics Shapes Drug Response
Pharmacogenomics in Cardiovascular Care
1. Antiplatelet Therapy: Clopidogrel and CYP2C19
Clopidogrel is a cornerstone antiplatelet drug, especially after procedures like percutaneous coronary intervention (PCI). However, its effectiveness depends on activation by the enzyme CYP2C19.
Certain CYP2C19 gene variants drastically reduce enzyme activity, meaning the drug never becomes fully “active.” Patients with these variants are termed poor metabolizers. Compared to normal metabolizers, they face significantly higher risks of adverse cardiovascular events, including stent thrombosis.
Alternative medications such as ticagrelor and prasugrel are not reliant on CYP2C19 for activation — making them preferable for patients with “loss-of-function” alleles.
2. Anticoagulants: Warfarin and DOACs
Warfarin requires a delicate balance — too little and clots form; too much and bleeding occurs. Genetics play a major role here. Variants in VKORC1 and CYP2C9 influence how individuals metabolize warfarin and determine the ideal dose.
When guided by PGx testing, warfarin dosing can be tailored, reducing harmful outcomes and improving anticoagulation control.
Direct oral anticoagulants (DOACs) like dabigatran also show genetic variability in how they’re metabolized, linked to enzymes like CES1 and transporters like ABCB1. Though less dependent on PGx than warfarin, emerging evidence suggests genetics still matter in individual response.
3. Statins: SLCO1B1 and Muscle Toxicity
Statins are among the most widely prescribed lipid-lowering medications, yet up to 20% of patients can experience muscle pain or myopathy. Genetic variants in the transporter gene SLCO1B1 are strongly associated with this risk.
A PGx-guided approach can identify patients at higher risk and inform safer drug choice or dosing, minimizing side effects without sacrificing cholesterol control.
4. Heart Failure and Genetic Modulation
In heart failure, beta-blockers and ACE inhibitors are foundational therapies. Genetic differences in ADRB1 and ACE genes have been observed to influence how patients respond to these treatments — from mortality benefits to symptom control.
By understanding these variants, clinicians can better customize therapy regimens that prioritize both safety and efficacy.
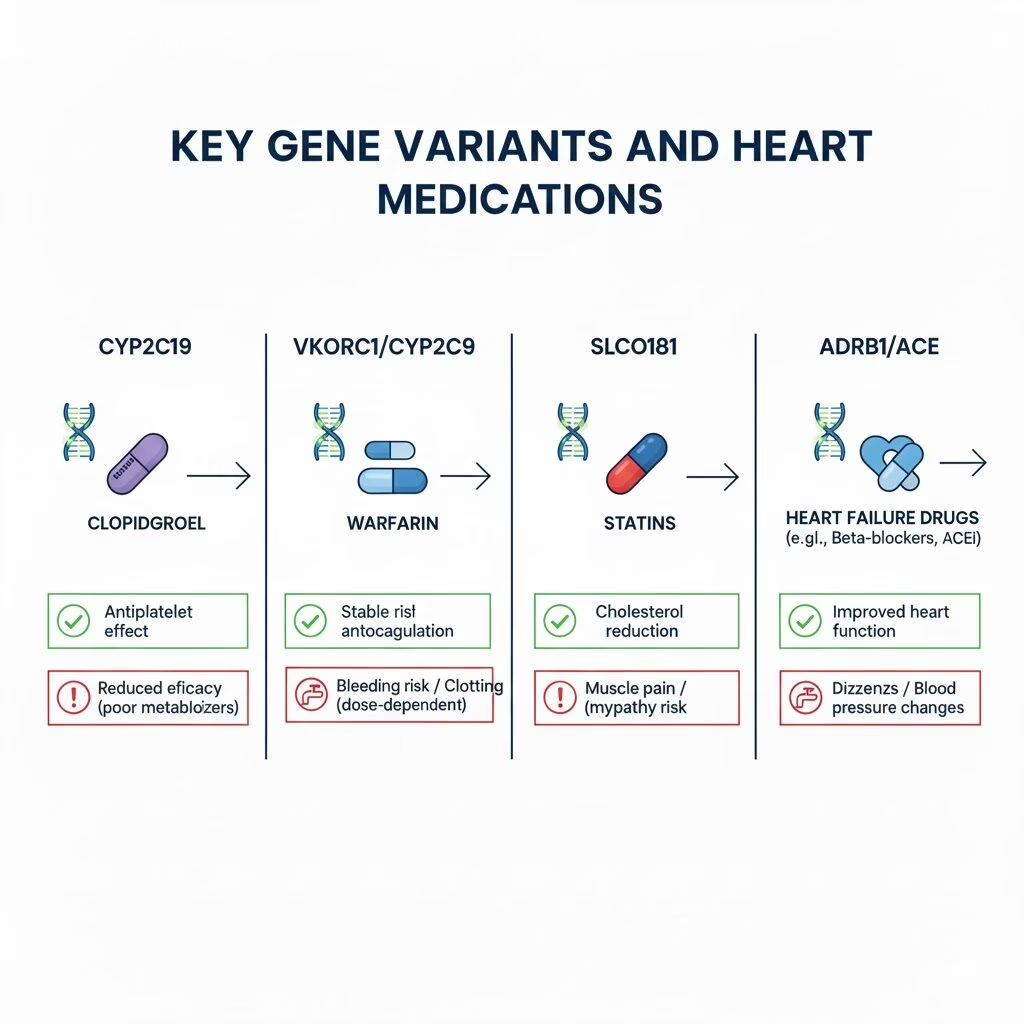
Key Gene Variants and Heart Medications
Why Pharmacogenomics Is Not Yet Routine Care
Despite compelling evidence, PGx is not universally standard practice in cardiology. Several barriers slow its widespread implementation:
1. Evidence and Interpretation Gaps
While some gene-drug pairs (e.g., CYP2C19–clopidogrel) have strong clinical evidence, others still lack definitive outcome data or consensus guidelines for routine use.
2. Cost and Accessibility
Genetic testing panels typically cost between $200-$500 and are not always covered by insurance, limiting access — especially in low-resource settings.
3. Clinical Expertise
Many clinicians lack training in genetic data interpretation, which slows adoption and reduces confidence in pharmacogenomics-guided decisions.
Read our blog on Clinicians in Saudi Arabia know about pharmacogenomics, new study finds.
4. Research Diversity Gap
Most pharmacogenomic data comes from European ancestry populations, leaving significant gaps in understanding genetic impacts in diverse groups, such as African, Asian, or Latino populations. This can inadvertently worsen health disparities if not addressed.
Ethical Considerations and Patient Perspectives
Pharmacogenomics introduces compelling ethical challenges. Consent, privacy, and data security are paramount when handling genetic information. Additionally, equitable access to testing and interpretation services must be ensured so that precision medicine doesn’t become synonymous with privileged care.
Merging Artificial Intelligence with Pharmacogenomics
While pharmacogenomics provides the biological foundation for personalized cardiovascular therapy, artificial intelligence offers the computational power needed to translate genetic insight into real-world clinical action. The convergence of AI and PGx has the potential to overcome many of the barriers currently limiting adoption and to do so at a scale that traditional approaches cannot achieve.
Cardiovascular pharmacogenomics generates complex, multidimensional data. A single prescribing decision may require consideration of multiple gene variants, comorbidities, concurrent medications, lab values, and clinical guidelines. AI excels in precisely this type of pattern recognition and data synthesis. Machine learning models can rapidly analyze genomic profiles alongside electronic health record data to generate clinically relevant recommendations, such as optimal drug choice, dosing adjustments, or risk alerts for adverse reactions.
One of the most promising applications lies in clinical decision support systems. AI-powered tools can embed pharmacogenomic insights directly into cardiology workflows, flagging actionable gene–drug interactions at the point of prescribing. Instead of requiring clinicians to manually interpret genetic reports, AI can translate results into clear, evidence-based guidance aligned with existing treatment protocols. This not only reduces cognitive burden but also increases clinician confidence in using PGx data.
AI also plays a critical role in expanding pharmacogenomics research. By analyzing large, diverse datasets, AI models can uncover novel gene–drug associations, identify population-specific variants, and refine risk prediction for underrepresented groups. This is particularly important for cardiovascular disease, where genetic diversity strongly influences drug response.
However, successful integration depends on high-quality data, transparent algorithms, and careful validation to avoid bias or over-reliance on automated outputs. When thoughtfully implemented, AI does not replace clinical judgment. Instead, it amplifies it, enabling pharmacogenomics to move from promise to practice in cardiovascular care.
Read our blog on 10 pharmacogenomic startups to watch.
Final Thoughts
Pharmacogenomics is more than genetic testing — it’s a blueprint for personalizing cardiovascular care. By harnessing the power of genes, clinicians can reduce adverse reactions, maximize therapeutic effects, and make smarter treatment decisions. While barriers remain, continued research, education, and infrastructure improvements are bringing personalized heart health ever closer to reality.